Study Background: The presence of minimal residual disease (MRD) after IC in AML patients is often a harbinger of relapse, and therapeutic targeting of MRD has emerged as an important strategy to improve long-term outcomes. The importance of the PD-1/PD-L1 pathway in immune evasion of AML has been demonstrated in murine AML models whereby PD-L1 and PD-1 were upregulated in leukemia cells, and PD-1 blockade suppresses invivo leukemia cell proliferation and improved survival in AML bearing mice (Zhang et al., 2009; Zhou et al., 2010). Combining IC with immune checkpoint inhibition (ICPI) has resulted in a significant clinical activity in some advanced solid malignancies including non-small-cell lung cancer. A recent single-arm phase 2 study combining IC with nivolumab showed the combination was feasible and resulted in undetectable MRD in 53% of patients with Complete response (CR)/CR with incomplete count recovery (CRi) (Ravandi et al, 2019). Here we report on the design of the first randomized multi-center clinical trial of IC+ICPI in fit AML patients.
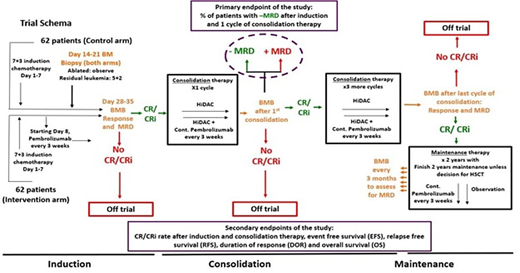
Methods: The primary objective of this investigator-initiated Cancer Therapy Evaluation Program (CTEP)-sponsored multi-institutional, randomized phase II study (NCT04214249) is to assess the percentage of patients with MRD negative CR (MRD- CR) as measured by flow cytometry at the end of first cycle of consolidation therapy with IC+ pembrolizumab and compare between the two study arms (Figure 1). Secondary objectives include rates of CR/CRi, complete remission with partial recovery count (CRh) and hematologic improvement (HI) to red blood cells and platelets, MRD negativity at Day 14, MRD-negative CR at end of induction therapy and MRD negative CR after last consolidation cycle, event free survival (EFS), relapse free survival (RFS), duration of response (DOR), and overall survival (OS), and proportion of patients who develop severe toxicity. Important exploratory objectives include MRD assessment by duplex sequencing (DS) and comparing DS and multiparameter flow cytometry for MRD detection.
The study will consist of an induction, consolidation, and maintenance phase of therapy. A total of 124 patients will be included (62 patients in the intervention arm and 62 patients in the control arm). Planned stratification factors include 1) age (younger than 65 vs. 65 and older), 2) cytogenetics by FISH or metaphase karyotype (presence vs. absence of core binding factor inversions and translocations), and 3) t-AML or AML arising from prior/antecedent MDS. All patients will receive cytarabine at 100 mg/m2/day continuous infusion on Days 1-7 and either idarubicin 12 mg/m2/day IV on Days 1-3 or daunorubicin 60 mg/m2/day IV on Days 1-3. on Day 8 of the induction chemotherapy, half of the patients will receive pembrolizumab IV (intervention arm), and continue Q3 weeks throughout Induction/Consolidation, and thereafter for up to 2 years from the start of maintenance. The other half will not receive pembrolizumab (control arm).
After 31 efficacy-evaluable patients (patients who had a bone marrow biopsy done after the first consolidation cycle) in each of the two study arms have had their primary endpoint results available, a formal futility and toxicity analysis will be conducted. Follow up will continue up to 2 years after the last patient is randomized.
Eligible patients are aged ≥18 and ≤75 years with newly diagnosed and pathologically-confirmed AML, including secondary AML arising from prior MDS and therapy-related AML. Patients with FLT3-mutated AML are excluded as well as those with prior allogeneic stem cell transplant, active infection requiring systemic therapy, and requiring use of high dose corticosteroids.
Responses in AML patients will be assessed using European Leukemia Net 2017 response criteria. EFS will be calculated as the time from initial treatment to either disease relapse or death. OS will be calculated from time from initial treatment to death. Biomarker analyses will include an extensive array of immunologic and correlative studies that will evaluate PD-1 and PD-L1 expression, immune cell subset analysis, leukemia specific T-cell response, cytokine level, T-cell receptor (TCR) repertoire, as well as RNA-seq, gut microbiome characterization and metabolomics, and whole exome sequencing for tumor and germline DNA.
Zeidan:Epizyme: Consultancy, Honoraria; Astellas: Consultancy, Honoraria; Daiichi Sankyo: Consultancy, Honoraria; Cardinal Health: Consultancy, Honoraria; Acceleron: Consultancy, Honoraria; Leukemia and Lymphoma Society: Other; Jazz: Consultancy, Honoraria; Celgene / BMS: Consultancy, Honoraria, Research Funding; Abbvie: Consultancy, Honoraria, Research Funding; Otsuka: Consultancy, Honoraria; Pfizer: Consultancy, Honoraria, Research Funding; Seattle Genetics: Consultancy, Honoraria; BeyondSpring: Consultancy, Honoraria; Cardiff Oncology: Consultancy, Honoraria, Other; Takeda: Consultancy, Honoraria, Research Funding; Ionis: Consultancy, Honoraria; MedImmune/Astrazeneca: Research Funding; Trovagene: Consultancy, Honoraria, Research Funding; Aprea: Research Funding; ADC Therapeutics: Research Funding; Astex: Research Funding; Incyte: Consultancy, Honoraria, Research Funding; Taiho: Consultancy, Honoraria; CCITLA: Other; Novartis: Consultancy, Honoraria, Research Funding; Boehringer-Ingelheim: Consultancy, Honoraria, Research Funding; Agios: Consultancy, Honoraria. Radich:Amgen: Consultancy; Bristol-Myers Squibb: Consultancy; Jazz: Consultancy; Novartis Pharmaceuticals Corporation: Consultancy, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal